 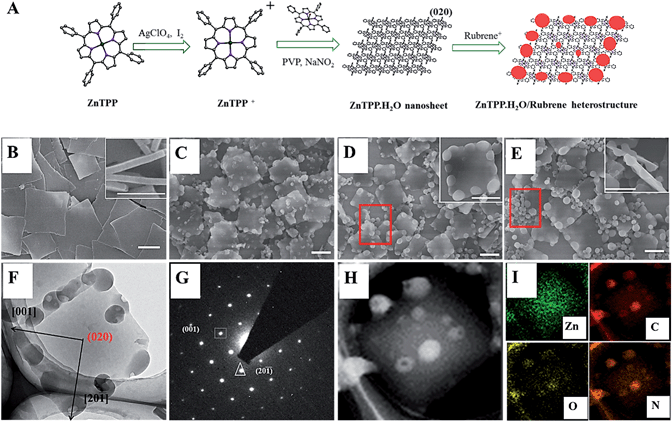
The other fatal problem of organic compounds is low conductivity, which will cause low-capacity utilization and poor rate performance. It cannot be ignored that the cation-selective membrane will hinder the ions transfer, which causes the unsatisfactory rate capability of C4Q. The cation-selective membrane is used to hinder the dissolution of C4Q. For instance, calixquinone (C4Q) has been reported in AZIBs and shows a high energy density, while its high solubility in aqueous electrolyte results in terrible cycling life. Unfortunately, the high solubility and poor conductivity of organic materials limit their development for AZIBs. Besides, the organic compounds are an alternative for AZIBs’ cathode due to many merits such as flexible structural designability, redox potential adjustability, and sustainability. Thus, that shortens the diffusion path and shows a capacitance-like behavior, which is different from inorganic materials based on ion insertion/de-insertion reaction process. Compared to the inorganic materials, the organic materials commonly display rapid reaction kinetics due to the rich exposed electroactive site on their surface. They suffer from sluggish ion diffusion kinetics and terrible structural stability due to the strong electrostatic effect between Zn 2+ and host materials. However, the inorganic materials have to tolerate repeating insertion/extraction of Zn 2+. For example, vanadium oxides, manganese compounds, and Prussian blue analogs are widely studied. To solve these issues, large amounts of inorganic compounds are reported over the last several decades. However, the energy density and cycling life of AZIBs are further limited by the lack of robust cathode materials. Compared to commercial lithium-ion batteries, aqueous zinc-ion batteries (AZIBs) possess enormous potential on large-scale energy storage devices due to many merits. The cutting-edge electrochemical performances of these novel compounds make them alternative electrode materials for Zn-organic batteries.ĭeveloping a durable and economical energy storage device is still a challenge for collecting energy from wind, solar, tide, and so on. Impressively, the battery can normally work at − 60 ☌ benefiting from the anti-freezing electrolyte and maintain a high discharge capacity of 201.7 mAh g −1, which is 86.2% of discharge capacity at 25 ☌. Additionally, the H +-storage mechanism of the 4S6Q compound is demonstrated by ex situ analyses and density functional theory calculations. This system displays a superlong life of 20,000 cycles with no capacity fading at 3 A g −1. 
The excellent rate capability is ascribed to the fast reaction kinetics. For instance, the Zn//4S6Q battery obtains 240 and 208.6 mAh g −1 of discharge capacity at 150 mA g −1 and 30 A g −1, respectively. Hence, the Zn//4S6Q and Zn//4S4Q batteries exhibit satisfactory electrochemical performance based on 3.5 mol L −1 (M) Zn(ClO 4) 2 electrolyte. The conjugated thioether (–S–) bonds as connected units not only improve the conductivity of compounds but also inhibit their dissolution by both extended π-conjugated plane and constructed flexible molecular skeleton. Because nitrogen in \(NH_3\) has an oxidation state of -3, it has the lowest oxidation state and will most likely be the reducing agent.Novel small sulfur heterocyclic quinones (6a,16a-dihydrobenzonaphthodithiinothianthrene-5,7,9,14,16,18-hexaone (4S6Q) and benzonaphthodithiinothianthrene-5,9,14,18-tetraone (4S4Q)) are developed by molecule structural design method and as cathode for aqueous zinc-organic batteries. Because \(NO_3^-\) has the highest oxidation number of +5, compared to the other molecules, it will most likely be the oxidizing agent. This is determined by comparing the oxidation numbers of nitrogen. \(NH_3\) is most likely to be a strong reducing agent. \(NO_3^-\) is most likely to be a strong oxidizing agent.The answer is C: In a redox reaction, there is always an oxidizing and reducing agent.When \(B\) gains electron, it is reduced, and is thus an oxidizing agent. When \(A\) loses electrons, it is oxidized, and is thus a reducing agent.The glucose is oxidized, so it is a reducing agent. Oxygen is reduced, so it is an oxidizing agent. The oxidizing agent is oxygen and the reducing agent is glucose. 
(aq)\) to an oxidation state of 0 in Cu(s). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
